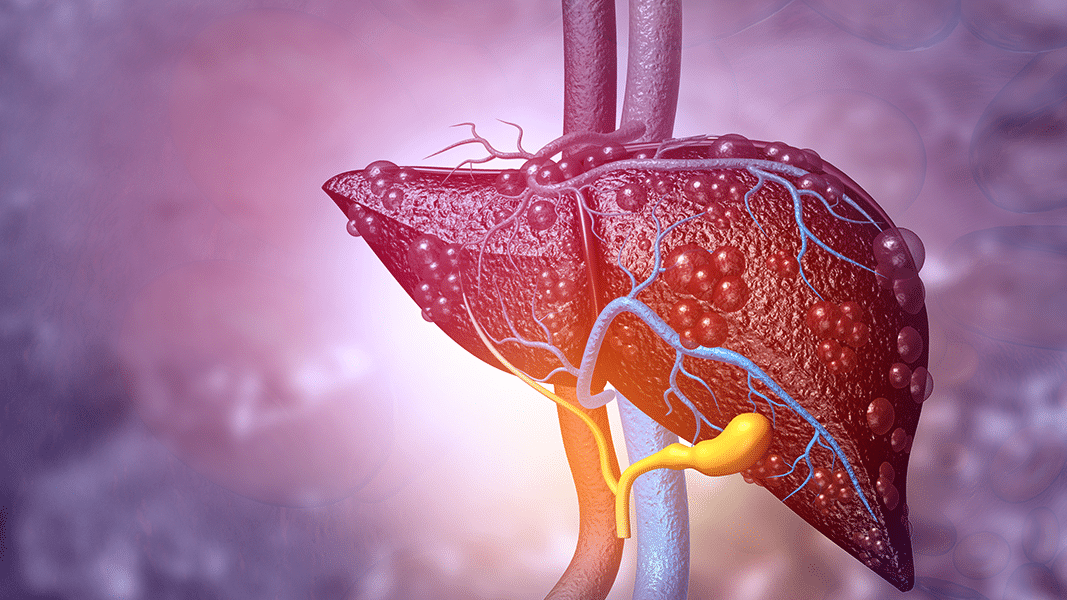
When the gene encoding FABP1 is deleted from rats, the rate of THC metabolism is dramatically slowed.
The endocannabinoid system is collection of proteins and oily lipids that orchestrate the body’s regulation of homeostasis. Most of the body is water, however, so cannabinoids usually reside on the boundary of cells. Transport molecules, like albumin, carry fats through the bloodstream. Within an individual cell, other molecules called fatty acid binding proteins (FABPs) carry cannabinoids to their destination. The destination could be CB1 receptors on the mitochondria or metabolic enzymes embedded in the cell’s manufacturing hub, the endoplasmic reticulum. A new paper from scientists at Stonybrook University examined the importance of FABPs in the breakdown of THC in the liver. THC normally activates CB1 and CB2 cannabinoid receptors on the cell surface, but it needs to be brought inside the cell to be metabolized. When the gene encoding FABP1 is deleted from rats, the rate of THC metabolism is dramatically slowed. The rats’ brains were exposed to about five fold more THC, in total. The maximal concentration of THC’s psychoactive metabolite, 11-OH-THC, was also diminished in the absence of FABP1. Acute doses of THC became more potent as a result. This could be one of the mechanisms by which CBD and THC interact — CBD competes with THC to be picked up by FABP1, consequently inhibiting THC’s breakdown.
Read study: FABP1 controls hepatic transport and biotransformation of THC
Adrian Devitt-Lee is a research scientist and longtime Project CBD contributor. © Copyright, Project CBD. May not be reprinted without permission.